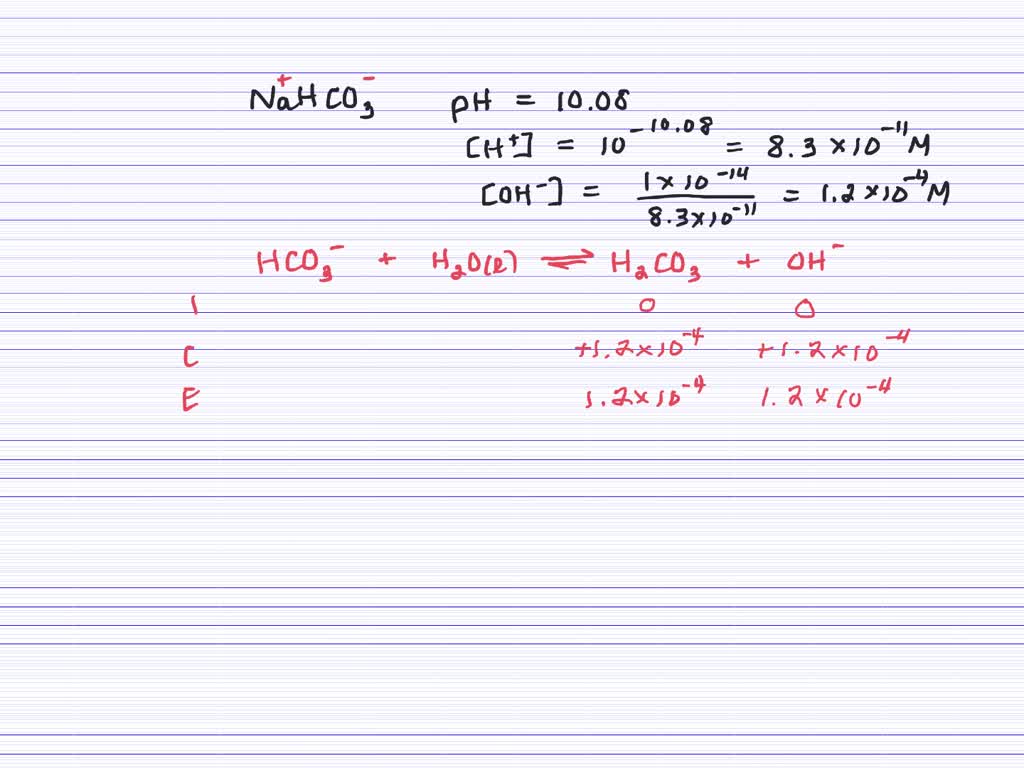
SOLVED:A solution of baking soda, NaHCO3, has a pH of 10.08 . What is the percent (by mass) of NaHCO3 in a 235-mL solution? (Assume a density of 1.00 g / mL.)

Preparation and properties of sodium bicarbonate‐incorporated LDPE films with deodorizing function for kimchi packaging - Jeong - 2017 - Packaging Technology and Science - Wiley Online Library

Simple Science: Vinegar & Baking Soda Balloon Experiment | Balloon science experiments, Science experiments kids preschool, Science experiments kids
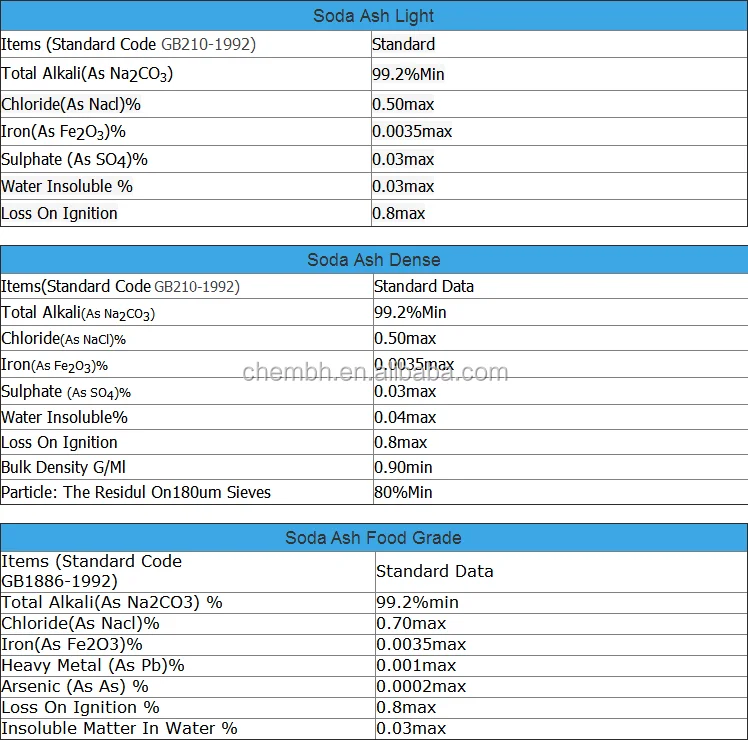
Hot Sale Chemical Formula Baking Soda Manufacturer / Factory Sodium Bicarbonate (nahco3) Price 144-55-8 - Buy Nahco3 Price,Sodium Bicarbonate (nahco3),Chemical Name Nahco3 Product on Alibaba.com

SOLVED: Determine the mass of baking soda (84 g/mol) needed to completely react with 355 mL of vinegar. Assume vinegar is 5% (m/m) acetic acid (60 g/mol) and the density of vinegar
![CERO ® SODIUM BICARBONATE 99% Pure [NaHCO3] CAS: 144-55-8 (200g) : Amazon.in: Industrial & Scientific CERO ® SODIUM BICARBONATE 99% Pure [NaHCO3] CAS: 144-55-8 (200g) : Amazon.in: Industrial & Scientific](https://m.media-amazon.com/images/I/81KKviHzabL._SL1500_.jpg)
















