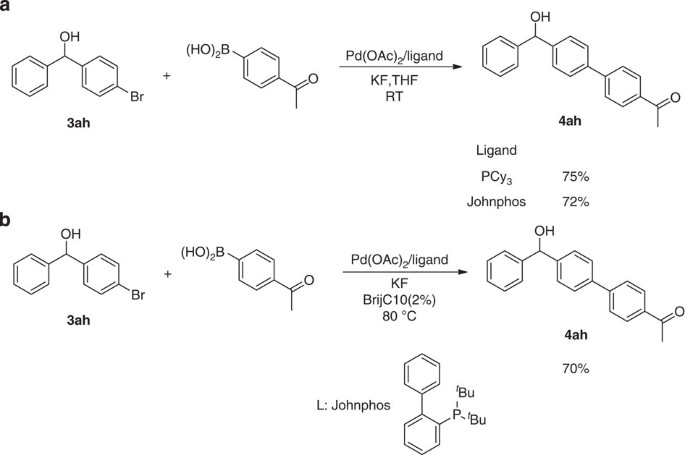
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
![PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/4e47c016d4cbb1df10c98e9abd9c1a88b9bc05b4/5-Figure1-1.png)
PDF] Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones† †Electronic supplementary information (ESI) available. See DOI: 10.1039/c8sc02038h | Semantic Scholar

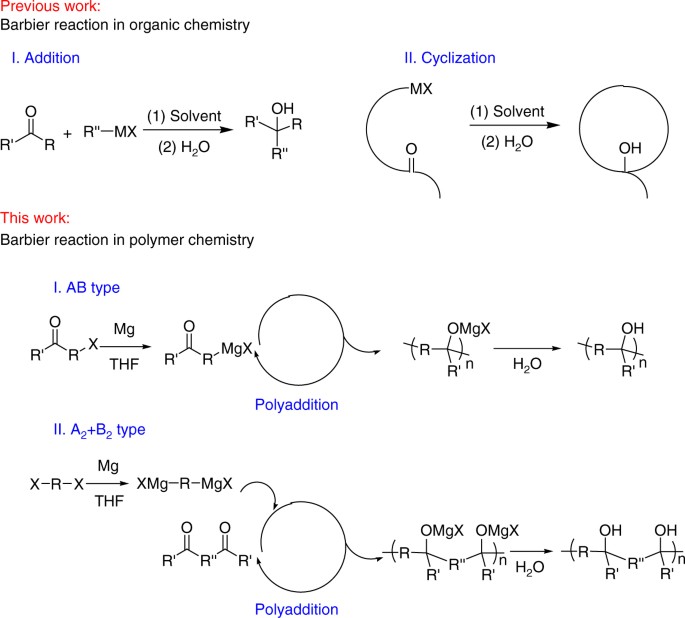
Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society
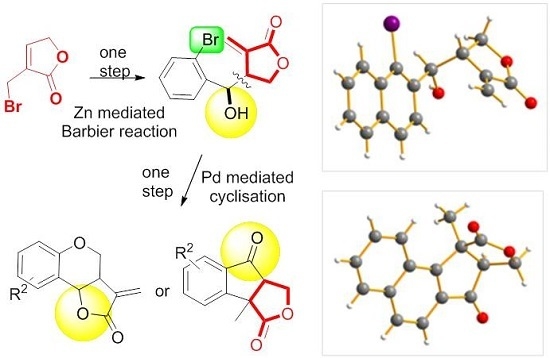
Molecules | Free Full-Text | From α-Bromomethylbutenolide to Fused Tri(Tetra) Cyclic Dihydrofurandiones through Barbier Reaction–Heck Arylation Sequence

Organozinc compound Barbier reaction Diethylzinc Organic chemistry, organozinc Compound, barbier Reaction png | PNGEgg
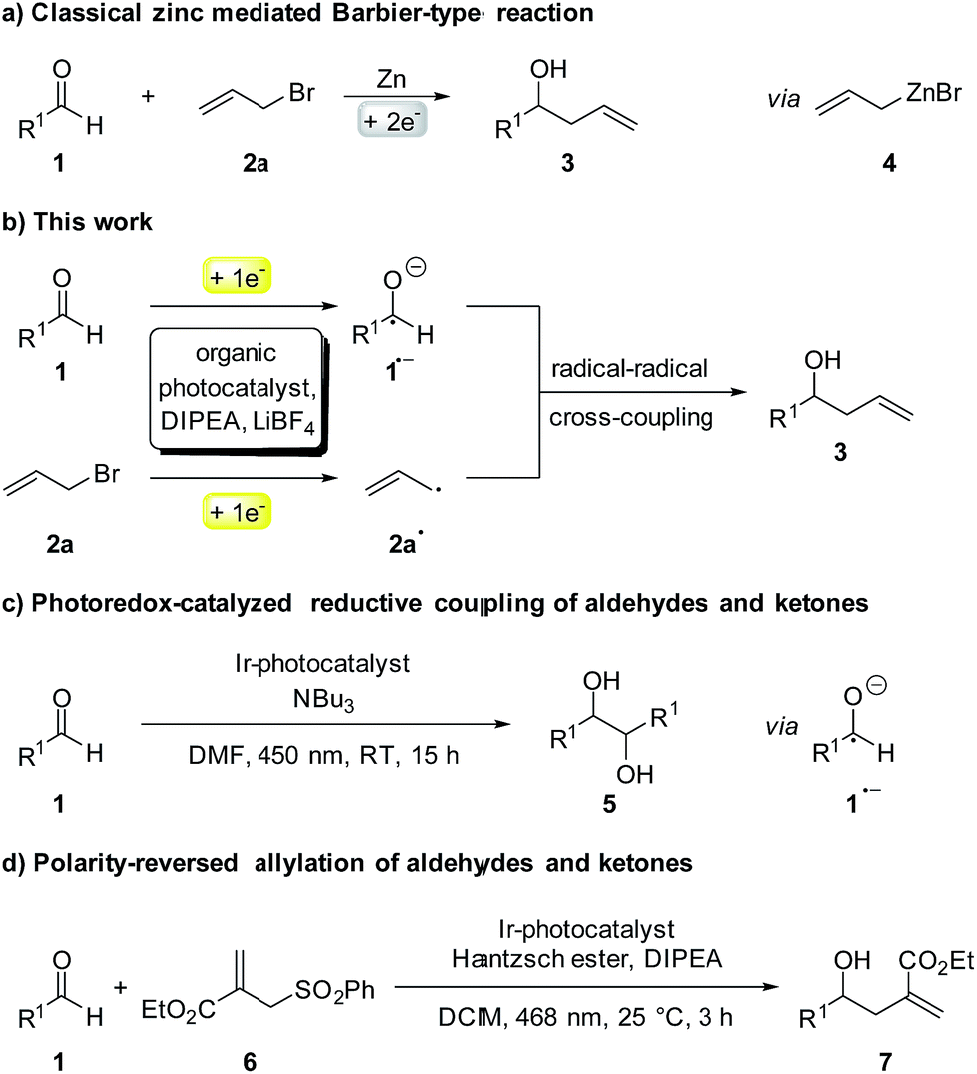
Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones - Chemical Science (RSC Publishing) DOI:10.1039/C8SC02038H

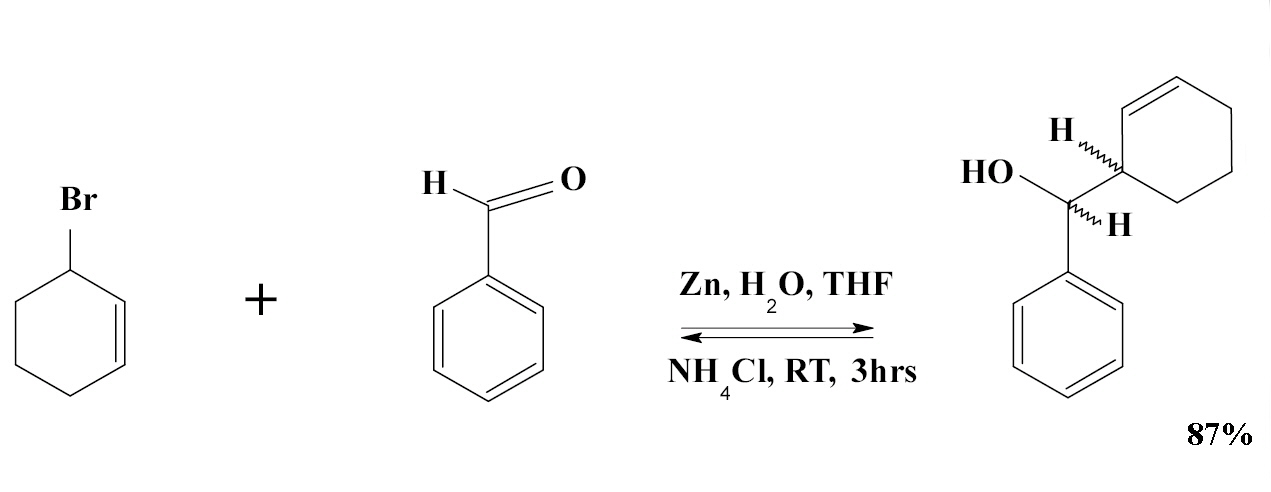
SOLVED: Znls) NH Cl(aq)THF Introduction: The Barbier reaction altemnative t0 thc Grignard reaction between organomagnesium halide and an aldehyde or ketone. Like the Grignard reaction , the product Is either secondary aleohol (
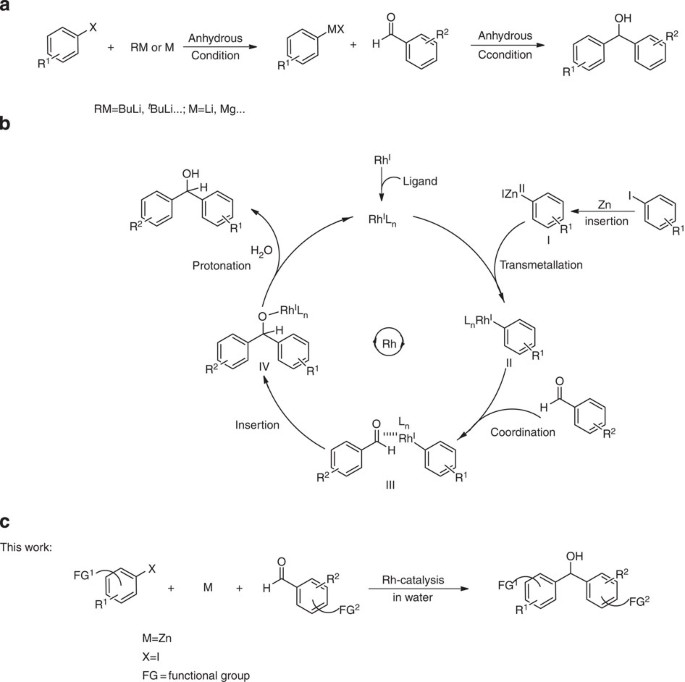
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
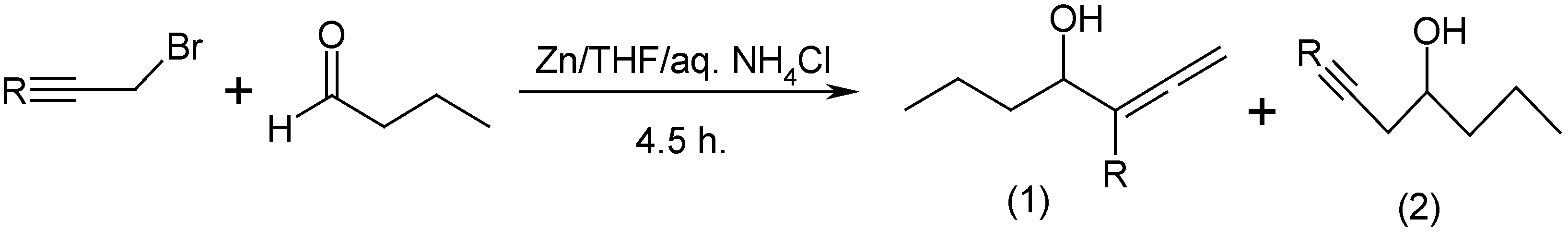
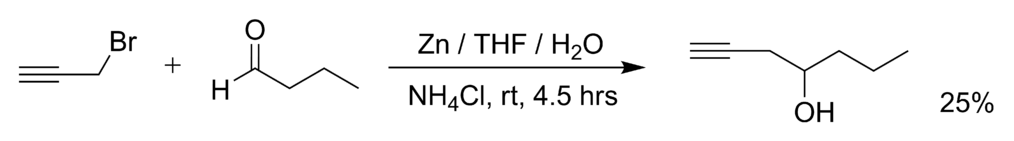
Molecules | Free Full-Text | Zn Mediated Regioselective Barbier Reaction of Propargylic Bromides in THF/aq. NH4Cl Solution | HTML