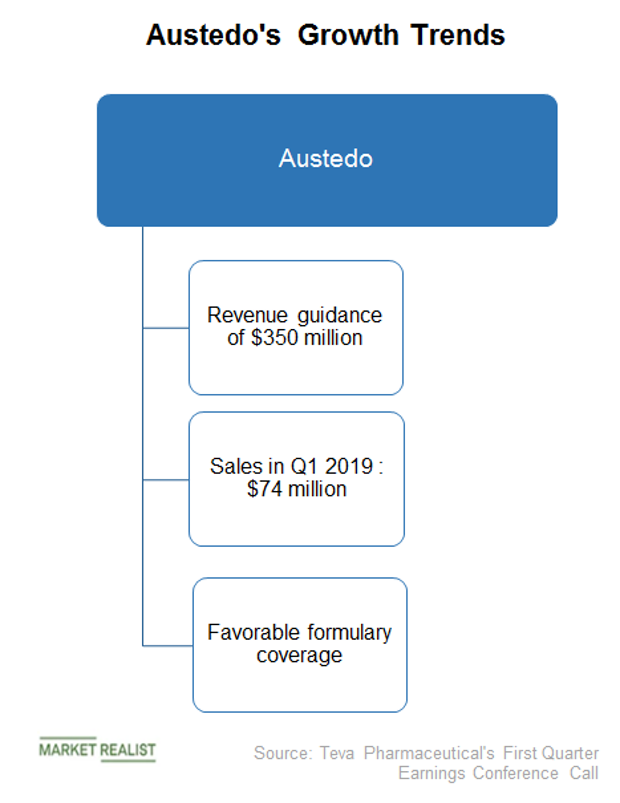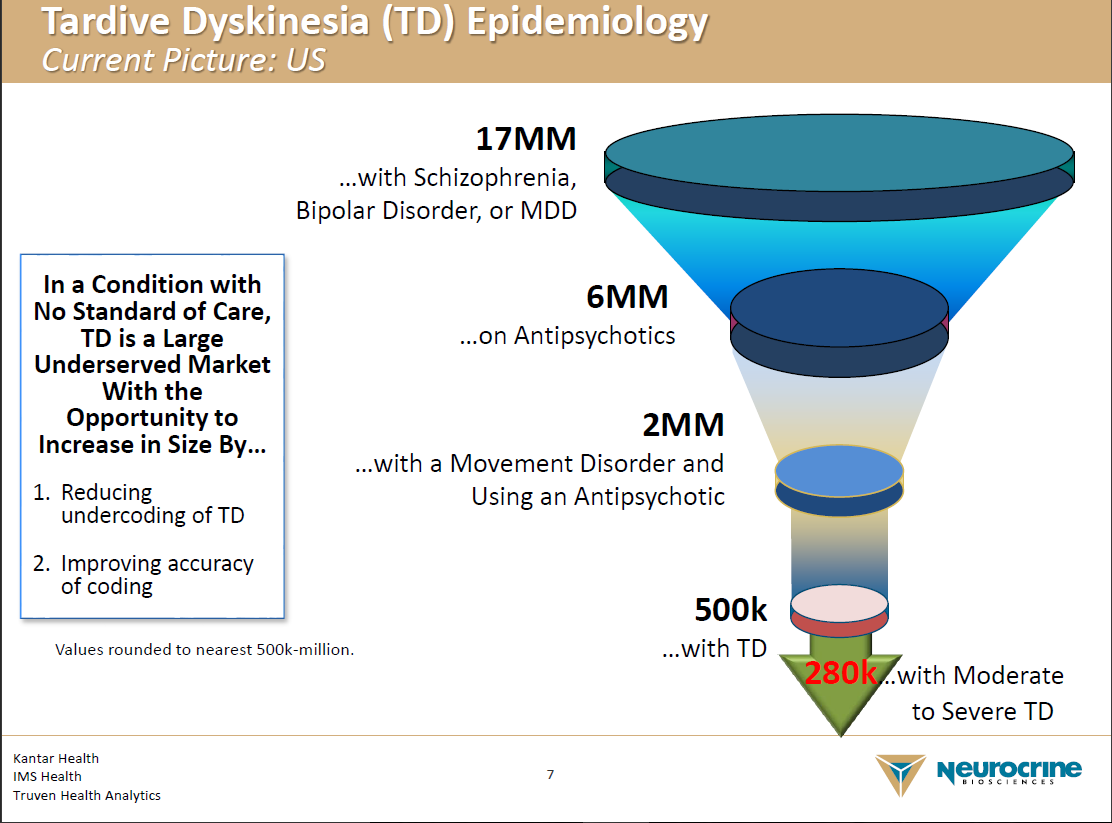
Teva: A Happy Parent To Tardive Dyskinesia Orphan Indication For Austedo (NYSE:TEVA) | Seeking Alpha
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena

Austedo - Deutetrabenezina 12mg caixa com 60 comprimidos - TEVA - ÁgilMed - Medicamentos Especiais e Nutrição Clínica
Austedo (deutetrabenazine) for the Treatment of Chorea Associated with Huntington's Disease - Clinical Trials Arena
Teva data indicates long-term benefit of Austedo for chorea associated with Huntington's | Seeking Alpha

Teva to Present New Data on Once-Daily AUSTEDO® XR (deutetrabenazine) Extended-Release Tablets at the 2023 American Academy of Neurology Annual Meeting | Business Wire